Cancer arises as a result of mutations and other genetic changes that shut down the control systems for growth that are normally present in our cells. New studies, however, emphasise the importance of the cancer cells’ communication with various cell types in the surrounding tissue, such as connective tissue, blood vessels and immune cells, in allowing the tumour to form, spread and resist treatment.
During the development of certain types of cancer, e.g. in the breast, liver and pancreas, the formation of connective tissue around and in the tumour is stimulated. However, researchers have not understood how the connective tissue affects tumour growth. Previous studies support the theory that it either drives the spread of the tumour by stimulating the formation of blood vessels, or that it weakens the immune system. However, inhibiting effects have also been studied, which could mean that the connective tissue encapsulates the tumour, thereby preventing it from spreading.
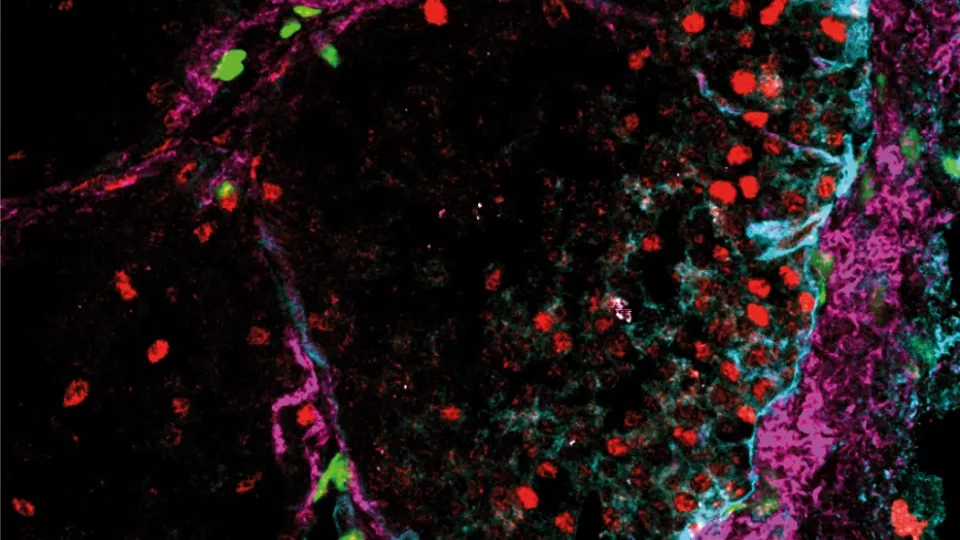
“We have investigated the pattern of gene expression in the connective tissue cells – known as fibroblasts – with a sensitive technique that enables the analysis of one cell at a time. By seeing which genes are active in 768 individual connective tissue cells from a mouse model of breast cancer, we were able to identify three different subgroups of fibroblasts which differ in function and origin”, says Kristian Pietras, research team leader at Lund University.
The largest group of connective tissue cells identified by the researchers controls the development of blood vessels; these cells are known as vascular fibroblasts. A second group of connective tissue cells has the task of producing connective tissue proteins that make the tumour stable and facilitate the migration of cells. These cells are called matrix fibroblasts and have been “kidnapped” by the tumour from the normal breast tissue as the tumour grows over them and converts them into helping. Finally, the researchers found a third group of cells, that are actually tumour cells that have disguised themselves as connective tissue cells.
“It is a well-known fact that tumour cells must undergo a transformation to acquire more connective tissue-like properties in order to be able to spread in the body – a process known as epithelial-mesenchymal transition. We were able to follow how, step by step, the malignant cells start to invade surrounding tissue. However, more detailed studies are needed to follow their journey all the way to a metastatic tumour in another organ”, says Michael Bartoschek, who is the principal author of the study.
Through precise tissue analyses, the researchers were able to confirm that the three types of fibroblasts are separate cell types which co-exist within different kinds of tumours. When they investigated the significance of the various subgroups of connective tissue cells for breast cancer prognosis, using tissue samples, they found that patients with large numbers of vascular fibroblasts or matrix fibroblasts in their tumours had a worse prognosis, as both these cell types affect the development of metastases.
Successful attempts to impede tumour cell communication with surrounding tissue through drugs already exist, but more research is required to find better treatment strategies targeting tumour cell communication, according to the researchers behind the study. The study confirms the old hypothesis that tumours have several different types of connective tissue cells, with different origins. The current findings open up the possibility of developing drugs that shut down specific functions in subgroups of connective tissue cells in order to achieve better therapeutic results with fewer side effects in breast cancer patients.
“We are convinced that more knowledge of the cellular structure of tumours and the function of communication between different cell types will enable us to find new ways to treat tumour diseases. In addition, measurements of the number of different connective tissue cells within a tumour can be developed to assess the risk of cancer recurrence in patients”, concludes Kristian Pietras.
The study was funded by grants from the European Research Council, the Swedish Cancer Society, the Swedish Research Council, BioCARE, Region Skåne, The Biltema Foundation, and Göran & Birgitta Grosskopf.
Publication: Spatially and functionally distinct subclasses of breast cancer-associated fibroblasts revealed by single cell RNA sequencing
Contact:
Kristian Pietras, Professor of Molecular Medicine, Division of Translational Cancer Research, Department of Laboratory Medicine, Lund University
+ 46 709 209 709
kristian [dot] pietras [at] med [dot] lu [dot] se (kristian[dot]pietras[at]med[dot]lu[dot]se)